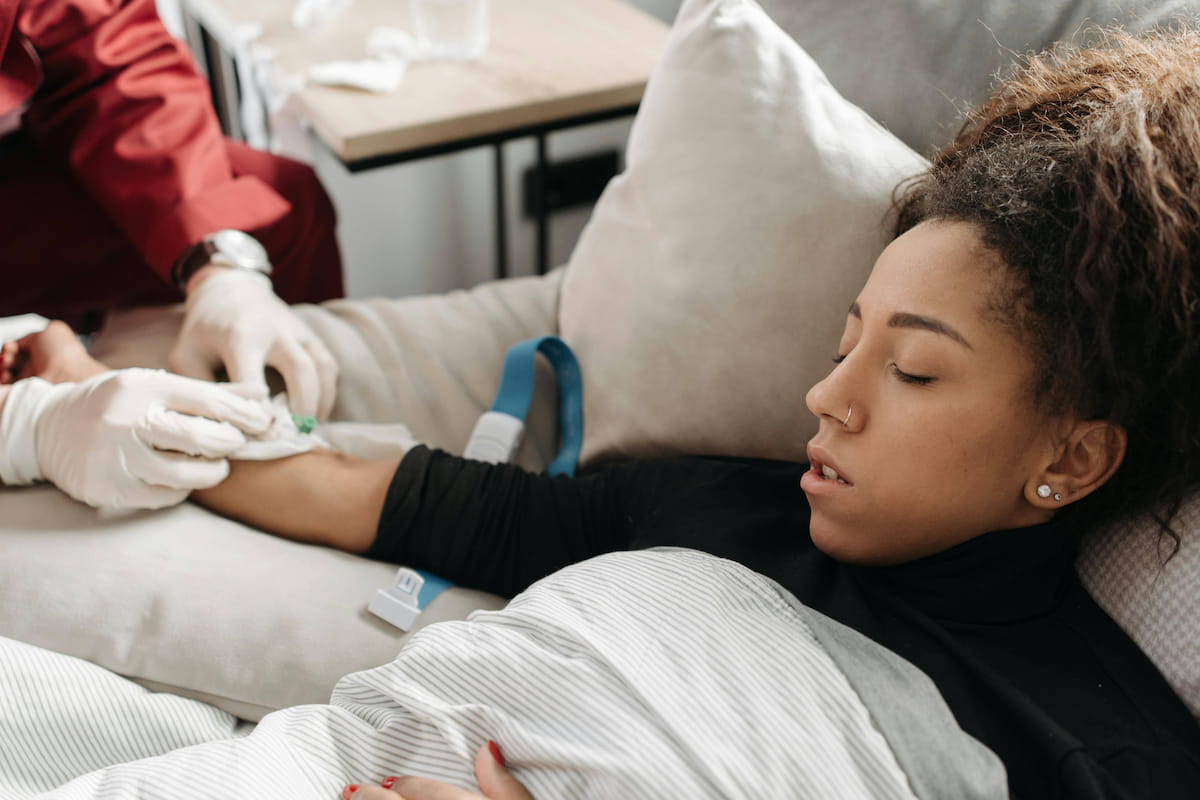
Cancer patients receiving chemotherapy spend hours in hospitals or care centers, biding their time while IVs drip tumor-killing chemicals into their veins.
But that might soon be a thing of the past for some patients, a new Mayo Clinic study says.
Chemotherapy can be safely delivered in patients’ homes, saving them a lot of hassle and drudgery, researchers reported in the April 2026 issue of the journal NEJM Catalyst.
“Cancer care has traditionally required patients to spend long hours in infusion centers, often far from home,” said lead researcher Dr. Roxana Dronca, site director of the Mayo Clinic Comprehensive Cancer Center in Jacksonville, Florida.
“This model allows us to safely bring high-quality care directly to patients, reducing burden while maintaining the standards patients expect from Mayo Clinic,” she said in a news release.
For the pilot study, researchers delivered 93 IV chemo infusions to 10 patients in their homes.
Patients maintained a continuous connection with their cancer care team through virtual visits and remote monitoring, researchers said.
Results indicated that home chemotherapy can be done safely and effectively, with no treatment-related infusion reactions or catheter-related infections among the patients.
Six of the 10 patients completed at least six months of home-based chemo, and four of those six asked to continue receiving chemo at home beyond the initial six-month period.
Four patients had to quit home-based chemo early, most because they needed a different chemotherapy regimen that couldn’t be delivered at their home, researchers said.
Most patients reported high satisfaction with their at-home care, and said they’d recommend it to others, according to the study.
Home-based chemo can reduce the physical, emotional and financial burdens of cancer treatment, researchers said. Patients don’t have to travel to a clinic for their chemo, and thus experience fewer disruptions to their daily life.
“This approach is about more than convenience,” Dronca said. “It’s about improving quality of life during treatment and expanding access to care for patients who may face barriers to reaching traditional cancer centers.”
Mayo Clinic is following up this pilot study with a full-fledged clinical trial, which launched in August 2023.
When it comes to cancer treatment, most patients need to travel back and forth to outpatient clinics repeatedly to receive care. For many in our community, they are more likely to face transportation challenges, limited access to local specialty care, work schedule inflexibility, and financial strain caring for their illness.
In addition, many loved ones and caregivers may take time off work to assist in transporting patients to and from appointments. Having at-home cancer treatment could reduce caregiver burnout and costs.
Ultimately, these barriers to consecutive, quality cancer treatment can greatly affect care. If at-home chemotherapy is approved and expands to more areas, it could likely reduce these burdens for our community.
Despite the name, at-home chemotherapy doesn’t mean you’ll handle treatment on your own.
At-home chemotherapy programs may involve:
It is important to note that not every chemotherapy regimen will qualify for at-home care, as some require immediate emergency support, complex monitoring, and specialized infusion equipment.
That being said, only certain patients and drugs may be suitable for at-home chemotherapy.
Absolutely. In fact, the Mayo Clinic has launched a larger randomized clinical trial, CARE Beyond Walls (Connected Access and Remote Expertise), to compare at-home chemotherapy with standard infusion care.
Clinical trials are crucial for advancing modern medicine — especially in treating diseases like cancer. Through clinical trials, researchers and healthcare providers can learn if a treatment is safe across all populations, if it improves quality of life, lowers costs, and increases access for underserved patients, such as the Black community.
Our community’s involvement in clinical trials is crucial, as we remain underrepresented in cancer research. Greater participation can help ensure treatments work across diverse populations.
It’s normal to have some questions and concerns about joining a clinical trial — it’s an experience that involves taking new drugs or treatments that could have potentially life-saving benefits, but can come with some unwanted side effects.
If you’re considering a cancer clinical trial, here are some questions you can ask researchers:

No, it is not yet widely available to the public. Most chemotherapy still takes place in an outpatient clinic or infusion center. Home-based programs like Mayo Clinic’s CARE Beyond Walls are still being studied and may only be offered at select health systems.
The good news is that many healthcare systems are exploring home-based care models thanks to the expansion of telehealth and remote monitoring in the past few years.
Going through chemotherapy is already challenging enough without the added burden of traffic, waiting rooms, and repeated visits to the clinic. While at-home chemotherapy is still being studied, early results are promising, with researchers suggesting that some patients may be safe and practical candidates for at-home treatment. If larger clinical trials confirm safety and efficacy, this care model could help bring cancer treatment closer to those who need it most.
More information
The American Cancer Society has more about chemotherapy.
SOURCES: Mayo Clinic, news release, March 19, 2026; NEJM Catalyst, April 2026

By subscribing, you consent to receive emails from BlackDoctor.com. You may unsubscribe at any time. Privacy Policy & Terms of Service.