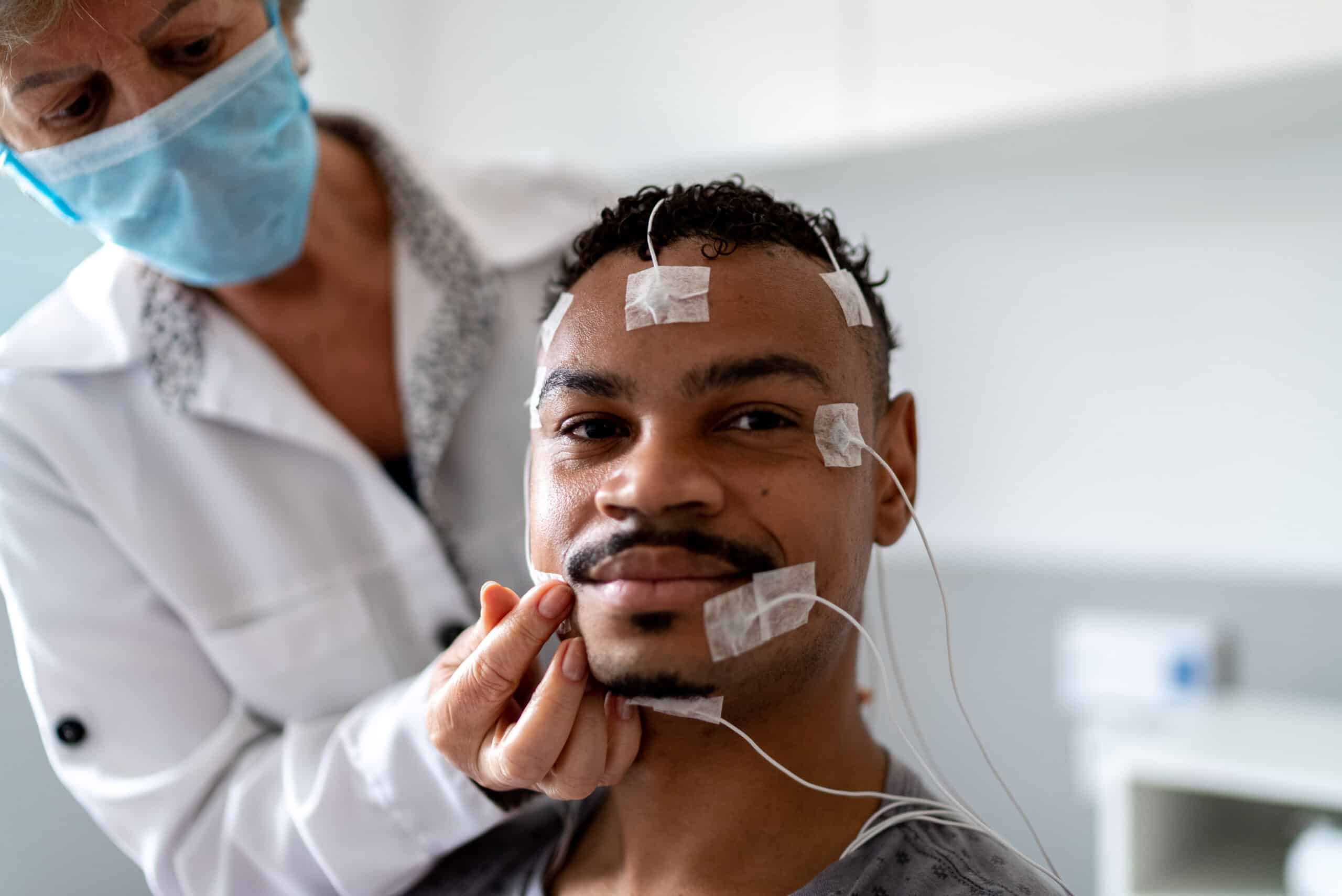
Clinical trials are research studies that use human volunteers to determine whether a medical treatment, drug, or device is safe for people to use. They aim to increase medical knowledge about disease treatment, diagnosis, and prevention through research. Clinical trials on humans are the final stage of the research process.
Although a doctor is usually at the forefront of each clinical study, the research team may include nurses, scientists, data experts, social workers, and other healthcare professionals. Clinical trials are commonly conducted at hospitals, medical centers, and universities — some clinical trials last days, while others continue for years.
Medical treatments don’t always work the same across populations, highlighting the need for diverse representation in clinical trials. The involvement of Black people in medical research is particularly important, as we need to ensure that new treatments are safe and effective for us. Participating in clinical trials can help improve outcomes across several diseases and conditions, such as heart disease, cancer, and diabetes.
Clinical trials consist of four phases:
In Phase I, researchers test a drug or treatment on a small group of 20-80 people for the first time. During this phase, the safety of a drug is studied, and any potential side effects are identified.
Phase I may take several months to complete. Approximately 70 percent of drugs studied during Phase I move on to Phase II.
In Phase II, a larger group of 100-300 people receives a drug or treatment to assess its effectiveness and safety further.
Phase II may last several months to two years — approximately 33 percent of drugs studied in Phase II move on to Phase III.
In Phase III, a drug or treatment is given to large groups of 1,000-3,000 people to confirm its effectiveness, monitor side effects, and compare it with similar treatments. Information that allows the new drug or treatment to be used safely is collected during this phase. Phase III testing may last one to four years, and approximately 25-30 percent of drugs tested during this phase move on to Phase IV.
In Phase IV, after a drug is approved by the U.S. Food and Drug Administration (FDA) and made available to the public, researchers track a drug’s safety amongst volunteers in the general population while seeking more information about optimal benefits and use.
Long-standing medical mistrust among Black communities due to incidents like the Tuskegee Syphilis experiment has made many of us feel as though the healthcare system doesn’t have our best interests in mind.
Fortunately, many safeguards have been put in place to protect participants in clinical trials, including Institutional Review Boards (IRBs), FDA oversight, and informed consent (the right to withdraw from research at any time).
RELATED: Are Clinical Trials Safe?

The clinical trial’s protocols define who is eligible to participate. Individuals may be included or excluded from a clinical trial based on their age, gender, health status, or medical conditions.
If you’d like to participate in a clinical trial, you should know the risks and potential benefits. When weighing the risks of participating in clinical research, you should consider the possible harms that could result from participating in the study.
Most clinical trials pose only a minor risk of discomfort. Still, the specific risks associated with a clinical research protocol will be described in detail in an informed consent document, which participants will consider and sign before participating in research.
A research team member will explain the study and answer any questions you might have about the study.
A well-executed clinical trial will allow you to access new research treatments before they are widely available, help others by contributing to research about new procedures, and receive professional medical attention from a research team of doctors and health professionals.
Here are some questions you can ask the research team before agreeing to participate in research:
If you want to explore clinical trials, but don’t know where to start, use the following tips:

By subscribing, you consent to receive emails from BlackDoctor.com. You may unsubscribe at any time. Privacy Policy & Terms of Service.